Since 1968, Gull Industries is the premier metal coating service provider throughout the central, southern region of North America. We pride ourselves as being a customer and market focused leader relative to customer services, product quality and environmental leadership. We apply 'Hi-Phos' E-Nickel at Gull Industries which is the most corrosion resistant version of all ENP processes available.

Cal-Aurum Industries provides metal plating and metal finishing systems to a variety of industries. We offer electroless nickel plating as well as gold, silver, rhodium, palladium, nickel, tin, tin/lead and copper plating. Our company formed in 1971, so we have years of experience and expertise to offer when we hear form you!

Since 1975, AOTCO Metal Finishing has been specializing in metal finishing systems and metal plating including tin, gold, copper, cadmium, chromium and electroless nickel plating. We are a metal finisher where quality and reliability count the most. Call us for more information.

More Nickel Coating Companies
Like other metal plating processes, nickel coating is used to create a protective layer on and alter the properties of the surface of other materials and parts. Nickel is a particularly valuable plating material due to its hardness and shinier finish. This means that it can be used to improve wear and corrosion resistance and can even be used for decorative purposes.
Nickel plating is generally accomplished in one of two ways. The first is through the electroplating process. Electroplating utilizes a solution of ionic nickel along with a negatively charged cathode and positively charged anode, usually comprised of the metal being plated. The ionic nickel is then supplied with electrons from the cathode which then produce a non-ionic metal film on the surface of the substrate or item of interest.
The other nickel plating method is called electroless nickel plating. This method of plating differs from the first in that it does not utilize the external electrical components, the anode and cathode. Instead this process utilizes a series of chemical reactions involving a reducing agent. These reactions cause the nickel to form the desired coating.
Of these two processes, electroless nickel plating is more commonly used due to the advantages it provides. This method generally results in a more even coating across the part’s surface and the coating can be evenly applied in surface recesses and in blind holes. This method also provides flexibility in the thickness of the plating. Regardless of the method used, nickel plating is a very useful and valuable process utilized in many industries. It is used in the aerospace, petrochemical, automotive, electronics, and commercial industries.








 Aluminum Anodizing
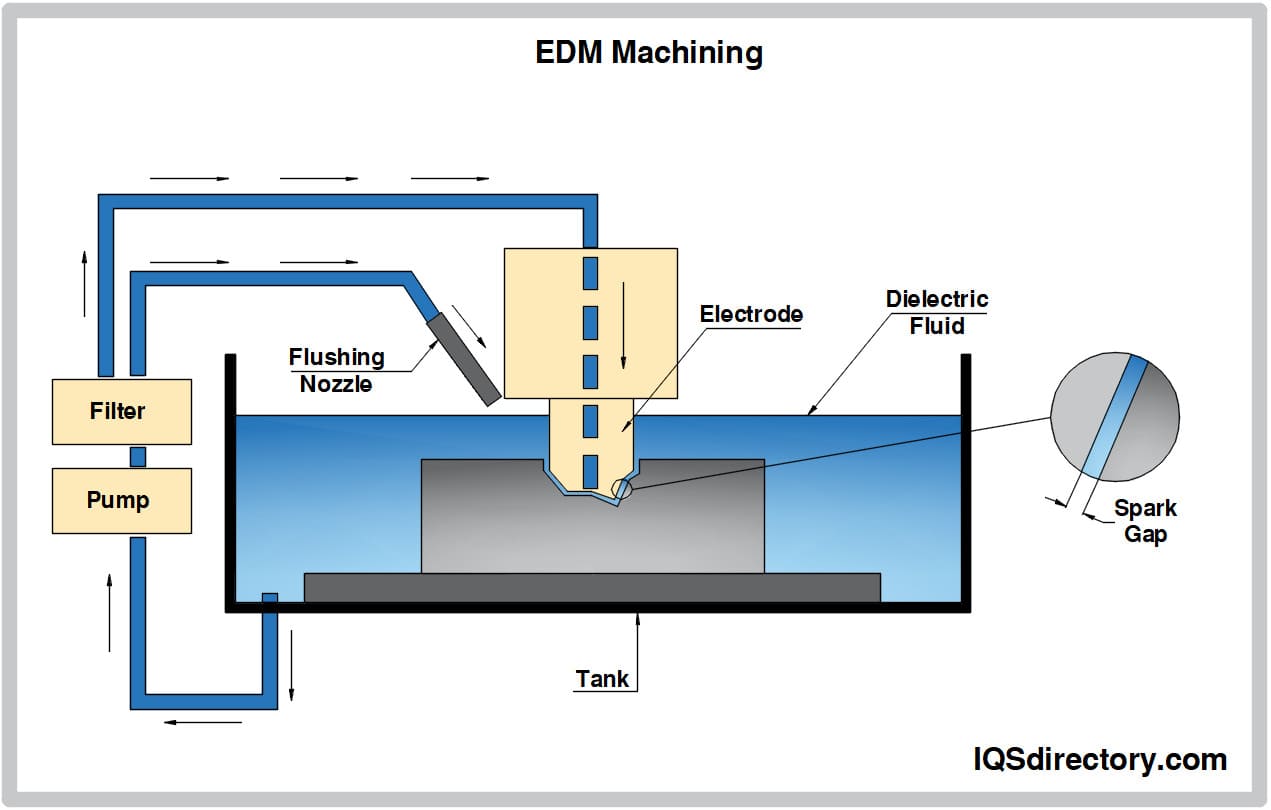
Aluminum Anodizing EDM
EDM Electroless Nickel Plating
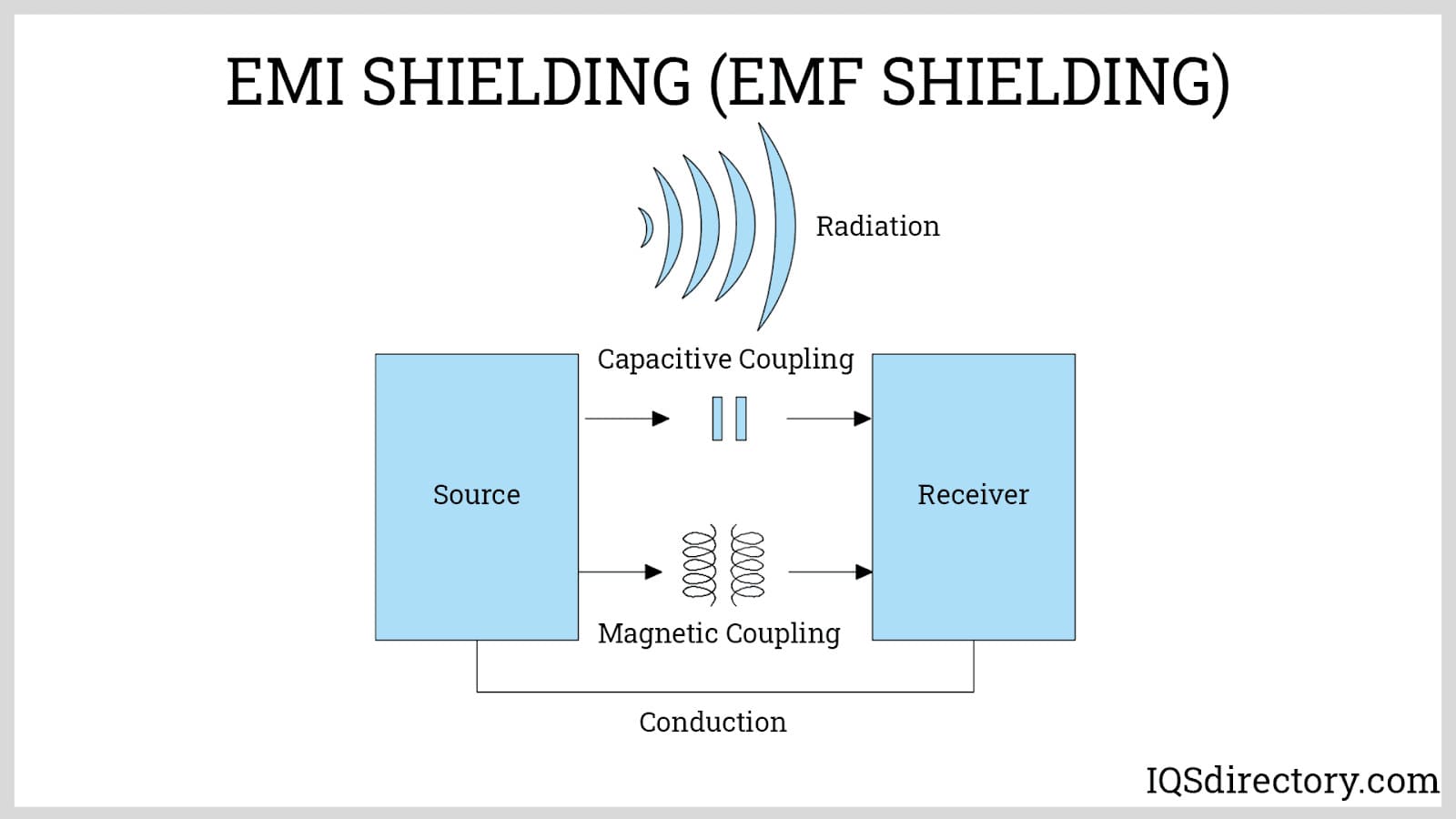
Electroless Nickel Plating EMI Shielding
EMI Shielding Heat Treating
Heat Treating Metal Coating Services
Metal Coating Services Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services