Since 1968, Gull Industries is the premier metal coating service provider throughout the central, southern region of North America. We pride ourselves as being a customer and market focused leader relative to customer services, product quality and environmental leadership. We apply 'Hi-Phos' E-Nickel at Gull Industries which is the most corrosion resistant version of all ENP processes available.

Cal-Aurum Industries provides metal plating and metal finishing systems to a variety of industries. We offer electroless nickel plating as well as gold, silver, rhodium, palladium, nickel, tin, tin/lead and copper plating. Our company formed in 1971, so we have years of experience and expertise to offer when we hear form you!

Since 1975, AOTCO Metal Finishing has been specializing in metal finishing systems and metal plating including tin, gold, copper, cadmium, chromium and electroless nickel plating. We are a metal finisher where quality and reliability count the most. Call us for more information.

More Electroless Plating Companies
As its name would suggest, this process does not utilize an electrical current like many other metal plating processes. Instead, this process requires a reducing agent. This chemical reacts with the metal ions and causes the metal to be deposited on the surface of a substrate or workpiece. The electroless plating process is most commonly used to deposit nickel alloys.
In order to utilize electroless plating, the substrate must be thoroughly cleaned in a pre-treatment process. The pre-treatment is a series of chemical cleanings which remove soils, debris, and other unwanted material from the surface of the substrate. Typically, the substrate must be rinsed multiple times between each chemical cleaning.
If this cleaning process fails to remove the debris and unwanted materials, the substrate cannot be plated properly. Once the plating process is completed, an anti-oxidation treatment is applied to the surface of the plated material followed by more water rinsing. Then the substrate is dried or baked to achieve the full hardness of the plating.
Though this process may seem very sensitive and involved, it offers many distinct advantages when compared with other plating processes. As stated before, electroless plating provides the benefit of not needing electrical power. This plating process also creates much more evenly coated surfaces than other processes and can evenly plate blind holes and recesses.
Many different finishes can also be achieved with electroless plating, including matte, semi bright, and bright finishes. Due to the wear resistance, corrosion resistance and hardness of the coatings, this process is utilized in many different industries and applications. Rotors, drive shafts, rails, valves, and mechanical tools are just a few examples of electross plated products.








 Aluminum Anodizing
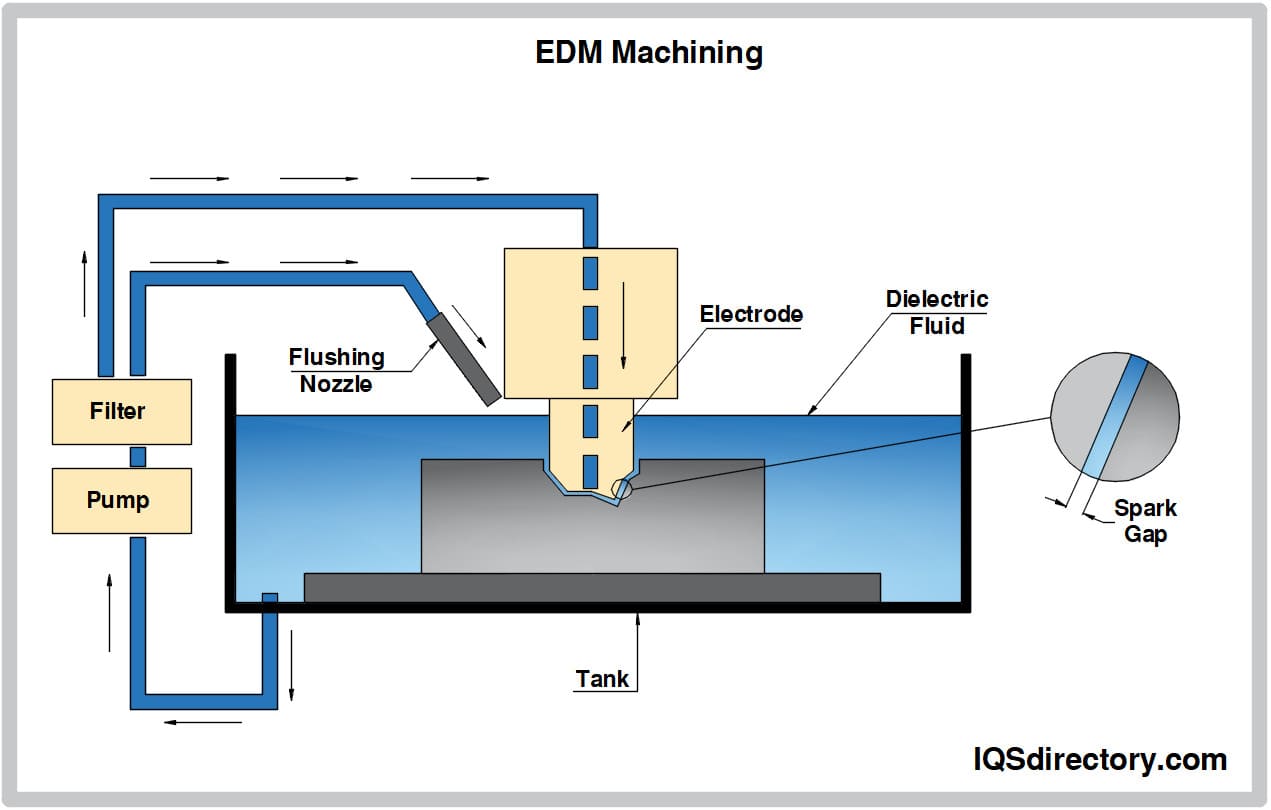
Aluminum Anodizing EDM
EDM Electroless Nickel Plating
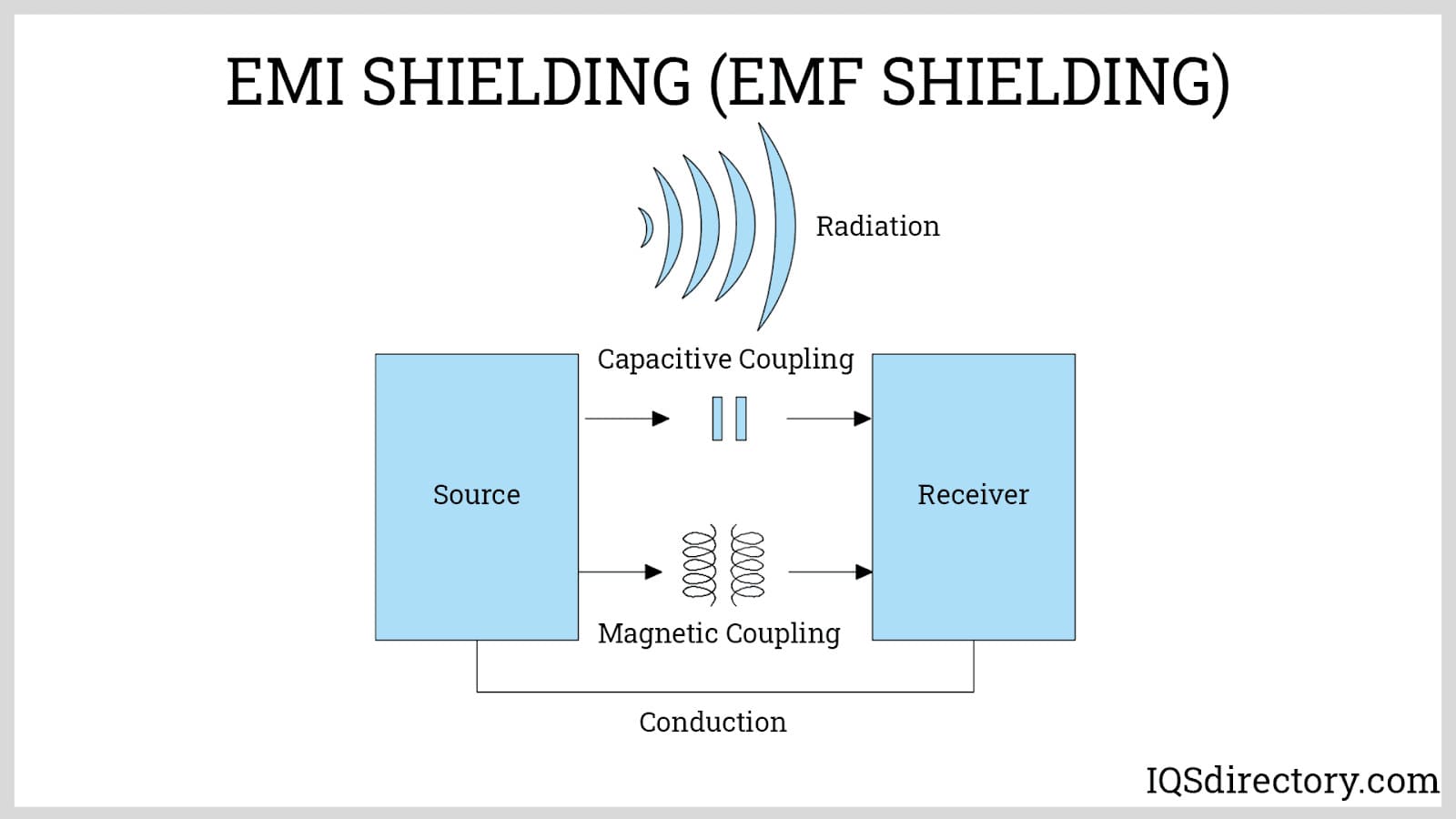
Electroless Nickel Plating EMI Shielding
EMI Shielding Heat Treating
Heat Treating Metal Coating Services
Metal Coating Services Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services